-
CIM PHARMA/MEDICO
Plug-and-play technology for serialization and inspection in the pharma industry

We ensure compliance to international standards
CIM delivers plug-and-play technology for serialization and inspection in the pharmaceutical industry. Our solutions are compatible with any pre-existing system, and compliance to all international standards is guaranteed. We ensure minimal impact on OEE with our module-based, easy-to-install, and easy-to-use solutions.
Serialization for pharma
Our serialization solutions guarantee full pharmaceutical traceability and integrate with any existing site server, line control or inspection system. Our solutions are based on pre-defined modules, and the need for customization is minimal. The result is a plug-and-play system that enables pharmaceutical companies to comply with all international serialization standards and distribute their products globally.
Inspection solutions for pharma
We deliver holistic machine vision systems and inspection solutions – all camera hardware and complete software-package included. Clients are ensured the right documentation, superior testing, complete validation, and full compliance-readiness to international standards.
In-line print
Integrate printing into your production line for reduced batch change-over-time, maximum flexibility, and improved quality control.
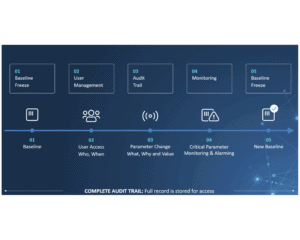
Keep Track and Stay Compliant
For production companies, keeping track of the activities on assembly lines and packaging lines is key to staying compliant with international standards and regulations. This is especially true for the pharma industry, where the CFR 21-part 11 regulation sets high standards for electronic documentation and signatures. Failure to comply means failure to operate, and that is devastating to business. So choosing the right audit trail system is more important than ever.
CIM TRACKS acts as a smart bridge between machine components at level 1 and the Enterprise System at level3 and allows you to monitor critical parameters and perform user management much more efficiently.
References
Don't take our word for it
Henrik Just
Project Manager Serialization
Stevanato Group
Jan Bigum
Lead Engineer
Grundfos Production IT
Andy Cumming
Consultant in Manufacture Change


Contact our specialist in IIoT Solutions
Anders Meister, Chief Sales Officer